Oxygen Is Necessary For Combustion %f0%9f%92%a8%f0%9f%8c%ac%ef%b8%8f Shorts Scienceexperiment
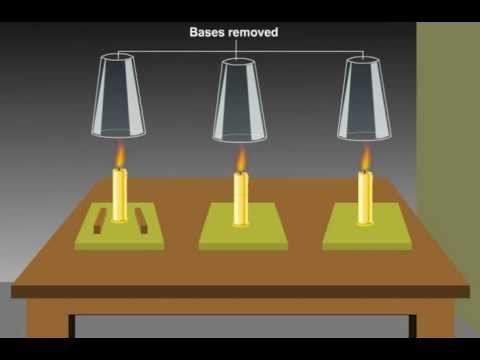
Combustion Why Is Oxygen Necessary For Combustion Class 8 Science Oxygen is required for combustion because it supports the chemical reaction in which a fuel burns to release heat and light. during combustion, oxygen reacts with the fuel, allowing it to oxidize and produce energy. without oxygen, burning cannot start or continue. Oxygen is fundamentally required for combustion because it acts as the primary oxidant in the chemical reaction. by definition, combustion is a rapid chemical process where a fuel reacts with an oxidant, typically oxygen, to release energy in the form of heat and light.

5 Combustion Requires Oxygen Stock Vectors And Vector Art Shutterstock Computes stoichiometric oxygen demand, required dry air, and the stoichiometric air–fuel ratio (afr) by mass and by moles. allows specification of excess air (λ) or equivalence ratio (ϕ) and returns the fully balanced products including any residual o 2. Discover why oxygen plays a crucial role in the combustion process and how it interacts with molecules during this essential chemical reaction. The stoichiometric air fuel ratio describes the exact quantity of oxygen required to consume every molecule of fuel. for example, the ideal theoretical ratio for gasoline is approximately 14.7 parts of air to 1 part of fuel by mass. Learn how to calculate oxygen requirements for fuel combustion in engines, boilers, & power plants. understand stoichiometric air & ultimate analysis.

Oxygen In Combustion Pptx The stoichiometric air fuel ratio describes the exact quantity of oxygen required to consume every molecule of fuel. for example, the ideal theoretical ratio for gasoline is approximately 14.7 parts of air to 1 part of fuel by mass. Learn how to calculate oxygen requirements for fuel combustion in engines, boilers, & power plants. understand stoichiometric air & ultimate analysis. Combustion reactions must have oxygen as a reactant. note that the water produced is in the gas state, rather than the liquid state, because of the high temperatures that accompany a combustion reaction. Oxygen is essential for survival and combustion reactions, including fire. this experiment explores the relationship between oxygen and fire, helping students learn about combustion. fuels such as natural gas, gasoline, coal, and wood require oxygen to burn. fire needs a heat source to ignite. The theoretically correct quantity of air or oxygen required to just exactly burn the fuel expressed as a ratio to the fuel burned, is called the stoichiometric ratio. Okay, let's break down how to determine the stoichiometric amount of oxygen needed for complete combustion. it's a bit more involved than a single number, as it depends entirely on what you're burning.

Solved Oxygen Heat And Fuel Are Each A Necessary Condition Chegg Combustion reactions must have oxygen as a reactant. note that the water produced is in the gas state, rather than the liquid state, because of the high temperatures that accompany a combustion reaction. Oxygen is essential for survival and combustion reactions, including fire. this experiment explores the relationship between oxygen and fire, helping students learn about combustion. fuels such as natural gas, gasoline, coal, and wood require oxygen to burn. fire needs a heat source to ignite. The theoretically correct quantity of air or oxygen required to just exactly burn the fuel expressed as a ratio to the fuel burned, is called the stoichiometric ratio. Okay, let's break down how to determine the stoichiometric amount of oxygen needed for complete combustion. it's a bit more involved than a single number, as it depends entirely on what you're burning.
Explain Why Oxygen Is Necessary For Combustion The theoretically correct quantity of air or oxygen required to just exactly burn the fuel expressed as a ratio to the fuel burned, is called the stoichiometric ratio. Okay, let's break down how to determine the stoichiometric amount of oxygen needed for complete combustion. it's a bit more involved than a single number, as it depends entirely on what you're burning.

Explain The Conditions Necessary For Combustion With Activities Cbse Tuts
Comments are closed.